« 2011 December | Main | 2011 October »
Wednesday, November 30, 2011
I found that fabrics dyed with blueberries faded the least. Can you please give me a scientific explanation why this happened?
Name: Stacey
—ADVERTISEMENTS—
Alum can be used safely as a mordant only if there is no risk that anyone will attempt to eat it.
  Not approved by the FDA for use in food; the fatal dose is one ounce for an adult (less than one small jar), and much less for a small child.
McCormick Alum Powder
Not approved by the FDA for use in food; the fatal dose is one ounce for an adult (less than one small jar), and much less for a small child.
McCormick Alum Powder
 
Turmeric works well as a natural dye, though it fades quickly in the light and cannot be expected to last.
  Simply Organic Ground Turmeric Root
Simply Organic Ground Turmeric Root
 
A good book is important for learning to use natural dyes.
 
The Art and Craft
of Natural Dyeing:
Traditional Recipes for Modern Use by Jim Liles
 
The Dyer's Companion contains clear recipes for mordants and dyeing.
  The Dyer's Companion (The Companion Series)
The Dyer's Companion (The Companion Series)
by Dagmar Klos
 
Natural Dye COLORS Collection

Natural Dye Kit
This comprehensive natural dyes kit contains Cochineal extract, Quebracho red, Logwood grey, Fustic, Indigo, Alum (Aluminum Acetate), Alum (Aluminum Sulfate), Cream of Tartar, Madder extract, Logwood purple, Cutch, Soda Ash, Iron, Scour (Washing agent for cellulose), Reducing agent for indigo (thiourea dioxide), Hide glue, pH strips, Mask and Gloves, and Instruction Book.
Buy from
Paradise Fibers

Country or region: USA
Message: Hi. I'm in 8th grade and I am working on a science project testing the fading of different plant based dyed fabric when washed. I found that fabrics dyed with blueberries faded the least. Can you please give me a scientific explanation why this happened? Also, fabrics dyed with gold beets based dye faded a lot more. Why is this? Please note that I was dying 95% cotton 5% spandex material. Also I did not use any mordants or fixatives to help hold the dye to the fabric. Thanks
What you have there, unfortunately, is not "dyed" fabric - it's "stained" fabric. You did not use any good dyes—there are other foods that will make much better dyes, especially if mordanted properly—and you didn't use any mordant that could help the better natural dyes perform well as dyes. I am sorry that you did not contact me (or read the 'schoolwork' section of my blog) before doing your experiment, for assistance in choosing a better question to test. You can use the results of the experiments you've done for your science project, but it might be better to write it up from a different perspective than you planned.
It looks like what you've been testing is, "Which food stains are the most difficult to remove?", rather than "Which plant dyes work the best?". In order to try to preserve what's left of your blueberry stain, you could look up "blueberry stain removal tips", which are widely available online, and then do the opposite. Since boiling water is effective at removing berry stains, avoid the use of any but cool water, if you wish to preserve your blueberry stains. (However, the oft-repeated advice to avoid "setting" the stain with the dry heat of a clothes dryer will not work well, in reverse, to make a stain act more like a dye.)
For any substance to be a dye, two things are required:- a good color; and
- an affinity for the fiber, so that the dye does not easily wash off.
Neither beets nor blueberries qualify as a dye under point 2. It's not enough for a substance to have a color; if it does not attach well to the fiber you're dyeing, it is not a dye.
Why did the stain from blueberries last longer than the stain from beets? In real life, it hardly matters, since it will still last almost no time at all! The bright blue color disappears almost immediately, replaced by a duller color, as the result of interactions with detergent or even simply the oxygen in air. Washing in sufficiently hot water, perhaps with the aid of an oxygen-containing laundry stain remover such as OxyBoost, will remove almost all that is left of the blueberries stain.
Blueberries are colored with natural anthocyanins (be sure to look up the chemical formula), which eventually wash out of clothing, and which change quickly from a lovely blue to a dull ugly color as the anthocyanin chemicals degrade. It appears that golden beets are colored with another natural chemical, betaxanthin (again, look up the chemical formula), but, as you have seen, it has no real affinity for unmordanted fabric, just like the betacyanins that color red beets.
Blueberries do have a slight affinity for the cotton fiber, but not a great one; this explains why they have formed more of a stain, although it's temporary, like the beets. Beets do not contain any usable textile dye at all, because their coloring truly does not have any affinity to the cotton fiber, not even enough to make much of a stain, as you saw.
The reason why boiling water is so good at removing blueberry stains, as well as any unattached dye of any sort, is that it significantly reduces the property called substantivity. Substantivity is the tendency of almost any large molecule to loosely associate with the textile fiber in question, such as cotton. Your results indicate that the pigment (not really a dye!) in blueberries has a greater substantivity toward cotton than the pigment in golden beets.
It is well known that neither blueberries nor beets make good dyes. There's quite a bit of information on this site available about this. Please read the following pages on my web site:
(Note that this last entry mentions post-mordanting, in which natural dyes can sometimes be saved by adding mordanting after the dyeing process, rather than before.)
which includes the statement "There is no point at all in tie-dyeing a sheet with blueberries or coffee unless you are going to preserve it, unwashed, as an art project. "
"Blueberries are colored with natural anthocyanins, while spinach is colored with chlorophyll. These colors are beautiful in the fresh food, but they turn dull when applied to textiles. For a thousand years, people have preferred to use indigo to dye clothing a blue color, because indigo is a much longer-lasting, brighter blue dye. To dye clothing or tapestries a green color, it is traditional to use indigo plus a natural yellow dye to make the green, because the green of chlorophyll that is found in the leaves of plants does not stay green, but instead turns brown."
Also see my explanation of exactly how to convert the colors of your test materials to numbers that can be graphed, using a scanner plus a photo program such as Adobe Photoshop Elements (which is far less expensive than the full Adobe Photoshop program, and also far less expensive than a proper scanning spectrophotometer, which is way beyond the reach of most middle and high school science programs). See:
Other science fair project questions and answers can be found in the "Schoolwork" section of my blog; click on the "Read More" under each title to see the complete article.
Reading a good book about natural dyes will be very helpful to you in the future before choosing natural dyes to try. I strongly recommend J. N. Liles' book, "The Art and Craft of Natural Dyeing", which contains a vast number of reliable recipes for dyeing not just wool, but also cotton, linen, and silk, using many different natural substances. Check your local public library to see whether they have it, or another good book about natural dyes and dyeing, in their collection.
You will need to cite this communication from me, among your references; here is one way to format this citation:
Burch, Paula E. "I Found That Fabrics Dyed with Blueberries Faded the Least. Can You Please Give Me a Scientific Explanation Why This Happened?" Paula Burch's All About Hand Dyeing. Paula Burch, 30 Nov. 2011. Web. 30 Nov. 2011. <http://www.pburch.net/dyeing/dyeblog/C22699859/E20111130074509/index.html>.
Tuesday, November 29, 2011
What dye can I use to cover stains on blends of cotton with modal, cotton with rayon, cotton with elastane, and viscose with elastane?
Name: Ruth
An ideal introduction to how to use synthetic dyes.
Country or region: USA, MA
Message: I need help. I am trying to find a dye appropriate for my clothes. They are a mix of cotton with modal, cotton with rayon, cotton with elastane, and viscose with elastane. These shirts were stained and I want to dye them to get rid of the stain. I want to dye them to their original color which is antique rose. Please help me. Thank you.
I can tell you what kind of dye is suitable for your clothing, but it's not going to cover up any stains. All dye is transparent. It will darken both the stained and the unstained regions of your clothing.
Applying an antique rose dye will have no effect on dark stains. If you have light stains on your clothing, as from bleach, they too won't be covered up well by dye, because the unstained areas get darker every time you dye the garment. (See "How can I fix bleach spots on clothing?".)
You might try Rit Color Remover on the stained garments. It will remove many stains, and it will also remove most but not all dyes, without destroying synthetic fibers as chlorine-based bleach does. Unfortunately, all color removers, other than chlorine bleach, work best in very hot water, but elastane is sensitive to heat, and is best treated with water only at or below 105°F.
A better answer might be to distract the eye from the stains by dyeing your pieces in a range of dark and light shades of rose, or in a variety of colors. A stain that is unwearably obvious on a light solid-colored garment may not matter much at all on a multicolored piece. See "How to Do Low Water Immersion Dyeing".
To answer the question of what dye is appropriate for your clothing, viscose is another name for rayon, and modal is a type of rayon; all of these fibers are reprocessed cellulose, generally made from wood. Since cotton is composed of cellulose, too, all three of these fibers are colored with the same types of dye. See "How to Dye Cotton" and "How to Dye Rayon". Elastane is the European name for what is usually called spandex or Lycra in the US; see "How to Dye Spandex (also known as Lycra® or elastane)". The best choice for a long-lasting dye on a cellulose fiber, or a mostly-cellulose spandex blend, is a fiber reactive dye, such as Procion MX dye. (See "About Fiber Reactive Dyes".) Unlike direct dye or all-purpose dye, this fiber reactive dye can be used at room temperature, which is much kinder to the elastane than the simmering to boiling temperatures required for the hot water dyes, and it is much easier to use than vat dyes.
You might be able to find fiber reactive dyes at a local crafts or fabric store in the form of Dylon Permanent Dye or Tulip Permanent Dye, though both require higher temperatures than Procion MX dye does. Since you're in Massachusetts, I strongly encourage you to buy from PRO Chemical & Dye, instead, an excellent dye supplier with very good prices that happens to be located in your area. Their PRO MX brand of Procion dye allows many more color choices than Dylon or Tulip, and it costs considerably less, per garment dyed, as well. See my page of "Sources for Dyeing Supplies Around the World", for links and contact information for these and many other dye sellers.
Monday, November 28, 2011
I would like to dye only the top of the quilting and leave the creases and thread white
Name: Kendra
Country or region: Colorado
Message: I have a queen size comforter, one large piece of white cotton with machine quilted designs. Assuming the thread is polyester (I have an OCD problem with tearing tags off), I would like to dye only the top of the quilting and leave the creases and thread white, or much lighter. I have not found a technique relatively close enough to achieve my desired look. Any suggestions? I thought possibly using gloves and lightly rubbing over the top. Would you have a dye type, brand preferance for this project? I would like an earthy dull tone blue and/or green.
The dye I recommend for your comforter is Procion MX type fiber reactive dye. You can buy it in Colorado, online if it's not nearby, from Colorado Wholesale Dye Company in Littleton, or, for a wider choice of pre-mixed colors, you can order dyes online from any retailer of Jacquard dyes, or from Dharma Trading Company in California, or PRO Chemical & Dye in Massachusetts.
I recommend fiber reactive dye because it lasts much longer than all-purpose dye, and it's also much easier to apply, since it's set with soda ash (the same chemical as in washing soda), rather than with heat the way all-purpose dye is. Setting with heat would be extremely inconvenient for an item the size of your comforter, plus it would be difficult to combine with the design you want.
Procion MX dye is the easiest to use of all the fiber reactive dyes, because its minimum reaction temperature, for fixing to the fabric, is 70°F. Other fiber reactive dyes require more warmth. Procion MX dye is also the cheapest of the fiber reactive dyes, in the US. We get very good prices compared to hand dyers in other countries.
It doesn't matter that you removed the tags from your comforter, as long as you're sure that it's cotton, because the tags never tell you the fiber content of the thread used to sew it together. There is a risk that the fabric might be 50% cotton and 50% polyester, in which case it will dye to a much paler color than 100% cotton will. 100% polyester would be much less practical to dye.
For your desired look, with the creases staying white (the thread will probably stay white no matter what you do), what I would do is buy a pre-mixed color of Procion MX type dye in a subtle earth-tone blue or green color that you like. It would be easiest to wait for warm weather so that you can do this outside without fearing splatters on your floor or furniture, but you can find a way to do it. Soak the comforter in a large bucket of soda ash mixed with water, one cup of soda ash for each gallon of water, then squeeze out as much of the extra liquid as you can, and spread the comforter out flat, using something like a plastic drop cloth underneath. Mix up the dye with water, following the same recipe as for tie-dyeing, then either put it into a spray bottle and spray dye across the comforter from an angle, holding a rag to catch any drips that fall from the sprayer, or use a large sponge to lightly apply splotches of dye where you want them. Spraying from a low angle keeps the dye from reaching inside the creases; it also leaves interesting shadows wherever there's a wrinkle in the fabric. Wearing gloves is something you should certainly do, but it will be easier to apply the dye lightly using a tool such as the spray bottle or a sponge. You don't want to squirt the dye directly on with a plastic yorker-top squeeze bottle, as we do in tie-dyeing, because it makes a very intense burst of color that will run into the creases. Air-brushing is another way to apply the dye lightly, but it requires a lot more equipment. You can cover the comforter with a other layer of plastic to keep it moist, or use urea in the mixture when you dissolve your dye in water. Keep the comforter in a warm place, minimum 70°F, overnight, since the reaction slows way down when the temperature falls below that.
Instead of pre-soaking the comforter in soda ash solution, you have another option of adding the soda ash directly to the dye mixtures, but if you do this, you must add the soda ash only when you are about to apply the dye. Be sure the dyes are completely dissolved before you add soda ash to them. If you mix the soda ash in with the dye before you are ready to use it, the dye will react with the water in the presence of the soda ash, and will no longer be able to react with the cotton of your comforter, so try to use the dye within half an hour of adding the soda ash. Use two teaspoons (or 10 ml) of soda ash powder for each quart (or liter) of dye mixture.
It's possible that you will end up with a little separation of colors on your comforter. Earth toned dyes are typically made by mixing together several different bright colors, such as blue with orange. From a distance the colors should blend together nicely. This can create quite a nice effect, but if it's something you want to avoid, do a test on something less important to you first. Some dye color mixtures tend to separate out a lot more than others; I have specific recommendations for how to mix colors to increase or decrease this effect, but I don't think you want to get into mixing your own colors yet, at this point. It's easier to select a premixed color from one of the major dye suppliers. If you don't see an earthy-enough blue or green, try mixing equal parts of a pre-mixed blue with a pre-mixed tan color; the yellowish character of a tan will turn a blue more greenish at the same time that it dulls it down to a more subtle color.
Be sure to prewash your comforter in the hottest water it can tolerate, to remove any invisible stains that might keep the dye from adhering evenly; sometimes spinning oils or other finishes are on the quilt when you buy it, so prewashing is essential even if it's new. After dyeing (you can wait a day or two if you like), wash the comforter, first in cool or lukewarm water to remove the soda ash and some extra dye, then in the hottest water available to remove all of the unattached excess dye. If the comforter can't tolerate hot water, then use warm water, and wash it multiple times.
(Please help support this web site. Thank you.)
Sunday, November 27, 2011
The dye rubs off when item is dry. How can I stop this?
Name: Julie
Country or region: UK
Message: Please I'd be very grateful if you could help. I have purchased an item that is 75% cotton and 15% mohair and the dye rubs off when item is dry. How can I stop this? Is it best to use a fixative or just wash repeatedly?
Crocking (rubbing off when dry) is a different problem from dye that runs when it's wet. It's harder to solve. You can't simply use a cationic dye fixative, such as Retayne, to fix the dye, because it won't work. The dye is rubbing off because some of it is not even loosely attached to the fiber.
There are two common causes for this problem. One occurs when the dyer neglects the all-important washing-out step after dyeing. It is always necessary to wash out excess unattached dye after dyeing. If this is the problem, then all you have to do is wash the item until it no longer crocks. Some people will tell you to wash it with salt or vinegar, but they don't know what they're talking about; neither will help any more than simply washing in water, though a little vinegar in the final rinse is good for restoring the pH of the mohair after any washing.
The other problem is caused by applying the dye the wrong way, with a total disregard for how the dye should be applied. This is a sign of an incompetent dyer. For example, when vat dyes, such as indigo, are applied with the wrong chemicals or at too heavy a concentration, the loose dye particles fail to penetrate the fiber and rub off easily afterwards. There is no solution at all for this one, if a few washings don't do it.
Any item that crocks dry dye should be returned to the seller for a full refund, because it was not dyed correctly. The seller cheated you by selling you an item that cannot be used without damaging whatever it touches.
If you can't return the item, try washing the item several times, and see if it helps. Often it will. If it does not, try soaking it in hot water. Unfortunately, while very hot water is the best for removing excess dye, it can also be bad for animal fibers such as mohair. When washing animal-hair products such as wool or mohair in hot water, do not subject the material to any sudden changes in temperature. Instead, gradually warm the water you are soaking the item in, and, afterwards, allow it to cool gradually.
For a small item that does not have to feel very soft, you can glue the loose dye particles in place by painting fairly heavily with a clear colorless fabric paint binder, such as Jacquard Products' Neopaque Flowable Extender or PRO Chemical & Dye's Lo Crock Binder. You can buy Jacquard's products online in the UK from George Weil. However, like any fabric paint, the clear fabric paint binder will leave a slightly harsher feel on the fabric, and it can be a lot of trouble to apply it evenly to a large piece of clothing, so it's not always suitable.
(Please help support this web site. Thank you.)
Wednesday, November 23, 2011
I am looking at starting a business dyeing ready-made clothing
Name: Pearl
Country or region: Dominica
Message: I am looking at starting a business, re dyeing of ready made clothing. I don't know much about dyeing "professionally" so I need to take a course. I am supposed to start a management course in January where I have to write the business plan and would want to do the dyeing course before.
I was about to shut down my PC when I came across your website, have been searching online trying to find a course available. Not sure if you do courses but would love your advice. What machines I should use etc, or maybe we could do an online course. I don't know I'm just asking cause I really need to get this done.Thank you, hoping to get a reply.
I'm afraid I have nothing to offer. My web site is all about hand dyeing, not professional dyeing.
I don't know anything about how to operate a professional dyeing shop. There seem to be very few to be found anywhere; the few I have found are listed on my page, "Where to Find a Custom Dyer". Be sure to study the contracts they have customers fill out before dyeing. Keep in mind that dye type and technique must be matched to the fiber content, and not all garments will survive dyeing.
The only machine I've ever used for dyeing is a top-loading washing machine. There are similar, larger-capacity machines that allow the use of water of any temperature, but I don't have any information about them.
(Please help support this web site. Thank you.)
Tuesday, November 22, 2011
What do you have to say about the presence of benzene in fibre reactive dyes?
Name: Carolynn
Country or region: Canada
Message: Paula, what do you have to say about the presence of benzene in fibre reactive dyes? I stopped using them thirty years ago after being warned about the carcinogenic nature of the dyes. I am very interested in trying them again but wondered if that warning is still valid. If so, how dangerous and what level of exposure are we talking about? I would not be making a career of dyeing yards of fabric! I am a fibre artist interested in making a little bit of hand dyed fabric for particular jobs. Thanks for your help.
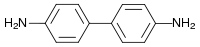 I have a suspicion that your real question is about a different chemical, benzidine, rather than with the chemical benzene. Benzidine is a dangerous chemical that has been found in other dyes, though never in Procion dyes, while benzene is not found in most dyes. I'll discuss both, as well as the question of whether Procion dyes are carcinogenic in general. I have a suspicion that your real question is about a different chemical, benzidine, rather than with the chemical benzene. Benzidine is a dangerous chemical that has been found in other dyes, though never in Procion dyes, while benzene is not found in most dyes. I'll discuss both, as well as the question of whether Procion dyes are carcinogenic in general.
It's ironic that sometimes you see warnings by the ignorant that reactive dyes are too dangerous to use because of carcinogenicity, when in fact it's the all-purpose dyes sold in grocery stores that used to be a danger in this respect. It's unfortunate that you were given such a warning thirty years ago, because it's simply not true that fiber reactive dyes are too dangerous to use safely. They are certainly safer than ordinary all-purpose dyes were, at the time you were given this warning.
By law, in the US, the Materials Safety Data Sheets (MSDS) must contain the names of all ingredients which are health hazards that are present at a concentration of 1% or greater, but they must list all carcinogens even if the are present at only 0.1% or greater. I don't know if the law in Canada is the same, but in practice it appears that the same MSDS pages are used in Canada as in the US.
If you look at an MSDS for benzene, such as the one provided by ScienceLab, you can see that benzene is classified as a proven human carcinogen by the IARC (International Agency for Research on Cancer). This means that any MSDS for a dye that contains as much as 0.1% benzene must list its presence.
For each dye you are concerned about, you should look up its MSDS. There are about a dozen Procion MX dyes in common use, used to mix all of the different colors of Procion MX dyes that are commercially available to us. Any retailer should, on request, supply you with the MSDS for any dye or other chemical they sell to you. Two easy places to look for MSDS information for Procion MX dyes are at Dharma Trading Company's MSDS index and at PRO Chemical & Dye's online catalog (see the link on the individual page for each dye color). Jacquard Products also provides MSDS pages, but they often seem less informative to me than some MSDS pages from other sources. You can also see MSDS for some Procion dyes at the chemical supplier Sigma Aldritch; their MSDS for reactive red 2 (red MX-5B) includes the statement that "This product does not contain any chemicals known to State of California to cause cancer, birth defects, or any other reproductive harm." It is clear from looking at any of the MSDS for Procion dyes that benzene is not a significant component of any of them.
In contrast, an MSDS for Direct Black 38, formerly an ingredient in many all-purpose dyes (all-purpose dyes include different brands such as Rit and Tintex), clearly states,
Carcinogenicity:
C.I. Direct Black 38 - California: carcinogen, initial date 1/1/88
NTP: Known carcinogen
IARC: Group 2A carcinogen (listed as Benzidine based dyes)."
Benzidine, rather than benzene, is a carcinogen of great interest in dye safety. Both benzene and benzidine are carcinogens, but there is little or no benzene in dyes, while there are many dyes actually based upon benzidine, or on either of two other specific chemicals, o-tolidine, and o-dianisidine. Dyes that are made from these chemicals may be contaminated with traces of the chemicals; worse, when absorbed into the body, they are changed by the body back into the base chemicals, benzidine, o-tolidine, and o-dianisidine. I strongly advise all of my readers to avoid all dyes that are based upon these substances.
Fortunately, Procion dyes have never contained benzidine-, o-tolidine, or o-dianisidine. No fiber reactive dye has ever contained any of these three dangerous chemicals. Unlike fiber reactive dyes, all-purpose dyes have in many cases been based upon benzidine. However, since the 1970s when they were in common use, all-purpose dyes that are based on benzidine have been phased out, and replaced by other dyes that are not based on benzidine. All-purpose dyes are now safer than they were in the nineteen-fifties, sixties, and seventies.
Unfortunately, while benzidine-based dyes have been phased out, there are still dyes now being sold, in the US, for use by children, that contain a o-dianisidine-based dye, in one brand of the "tie dye cords" that are so inferior to Procion dyes. I always recommended against these cords because of the poor quality of the dye contained in them, but I particularly recommend against using the ones that contain o-dianisidine. They can be used safely by careful adults, but, in my opinion, are too likely to be used unsafely by children, who are the target audience. (See "o-dianisidine in dye-impregnated tie-dye cords".)
We don't know that any particular textile dye is safe to eat, of course. Only food dyes have been tested for safety with truly careless use. Sometimes a food dye which has been allowed in the food supply for decades will be banned due to new evidence that it might have a small tendency to cause cancer, in a very small fraction of the people who eat it. Since textile dyes have not been tested in this way, it's always possible that one might turn out to be carcinogenic when eaten; this doesn't matter much to us, since we're not going to be eating any of them. We should always be careful, with any household chemical, to treat it as though it is a hazard. Wear gloves to prevent it from getting on your skin, wear a dust mask and be careful when working with dust powder, so that you don't breathe it, and wipe up any spills immediately.
There are other dyes that are much more dangerous to work with than Procion MX dyes. Some of the components used in naphthol dyes, for example, can be absorbed through intact skin when spilled on it, in contrast to Procion dyes, which react with the dead cells that serve as a protective layer on our skin, and are not absorbed into living cells.
The true, known hazard with Procion MX dyes, and any fiber reactive dye, is that breathing the dye powder can cause respiratory allergies, similar to the dust allergies many of us have, but with potentially worse results. If you become allergic to a particular type of dye powder, then, when you continue to use it, you are likely to have an asthmatic reaction. These sorts of allergies are not uncommon in the textile industry; they are much rarer among dye artists who follow the recommended precautions. Once you have developed an allergy to a dye powder, you must quit using it, and substitute a different kind of dye (for example, using Drimarene K fiber reactive dyes instead of Procion MX fiber reactive dyes). It is better to take careful precautions from the beginning, so that the allergy never gets started.
Looking at Sigma Aldritch's MSDS for reactive orange 4 (orange MX-2R), you see repeated statements, attributed to various agencies, that "No component of this product present at levels greater than or equal to 0.1% is identified as probable, possible or confirmed human carcinogen". However, that doesn't mean that it's safe to spill dyes, get them on your hands or in your eyes, or allow them to dust up into the air so that you and others breathe them. This is true for any dye, not just fiber reactive dyes, and in fact is true for most household chemicals. The MSDS contains these warnings:
- Inhalation: May be harmful if inhaled. Causes respiratory tract irritation.
- Skin: May be harmful if absorbed through skin. Causes skin irritation.
- Eyes: Causes eye irritation.
- Ingestion: May be harmful if swallowed.
All of these potential dangers can be safely managed by taking care to wear a dust mask, wear gloves and (if necessary) safety glasses, and avoid doing anything that might cause you to breathe or eat any of the dye. That last sounds a little stupid, but I've many times seen artists eating a snack or smoking cigarettes while painting with oils; obviously that sort of carelessness is easy to avoid, and incredibly foolish to engage in. Of course, cigarette smoking is a particular hazard for bladder cancer, as it greatly multiplies the danger of benzidine-based dyes, even for someone who is reasonable about hand-washing; the use of benzidine dyes is more dangerous in people who also smoke. It's remarkable, though, just how much more careless than necessary some people choose to be. I've seen artists who didn't take even the most obvious precaution of not using hands covered with fresh lead-, cadmium-, and chromium-based oil paints to handle something they're putting in their mouths. I recall reading a description one supposed dye company employee (of a company that is no longer extant) of never bothering to wear gloves or a dust mask while measuring out dyes, and ending up breathing so much dye that it appeared in his handkerchief when he blew his nose.
Procion MX dyes are safe to use, as long as you are careful and follow the usual rules of safety for household chemicals. They are no less safe than many household chemicals people don't think much about, such as dishwasher detergents, and they are safer than many.
Also see:
Answers to questions about dye safety in this blog
Saturday, November 19, 2011
I want to purchase a beaded taffeta dress and dye it for my daughter's wedding
Name: Thea
Country or region: Texas
Message: I am wanting to purchase a dress and dye it black for my daughter's wedding. It is made of iridescent taffeta. It does say dry clean only. There is beading on it, so I am certain that is why it is dry clean only. I am not worried about the iridescent part, I am worried about not wearing the right color for her wedding. Thank you.
What could possibly go wrong? Let's go through it step by step.
First, you have to pre-wash the dress, before you can dye it. Some dresses marked "dry clean only" do survive washing, but those with beading or sequins, and those with linings, are more likely to be permanently damaged. The beading may be sewn on or glued on. If it's sewn, a few of the beads may break in the process of washing; if it's glued, then the beads may become unglued. If there is a lining, it is apt to shrink to a different extent than the outer layer, resulting in deformation of the garment's shape, so that the dress no longer lies correctly.
Assuming that you've successfully washed the dress, and that it has escaped any significant damage, then you need to choose the right dye for the fabric. Check the fiber content. Taffeta is a word that describes the way the fabric is woven, but it doesn't tell you anything about what it's made of. You would probably have been more specific if your dress were made of silk taffeta, though, instead of a synthetic fiber such as polyester, acetate, or nylon. If your taffeta is made of polyester or acetate, then the only way you can dye it is to boil it in a very large cooking pot with a special dye for synthetic fibers, which is a type of dye called disperse dye. Cotton and silk can be dyed at room temperature in a plastic bucket, or in the washing machine, but polyester or acetate cannot.
The next problem is acquiring the cooking pot to do your dyeing in. If it's a short sleeveless dress, then you won't need as large a pot as you will if it is a long dress with sleeves. The cooking pot needs to be large enough for the dress to move freely in the dye mixture in the pot, as you stir it. Estimate how much the dress weighs, while dry, by weighing a similarly-sized dress. If your dress weighs one pound, then, depending on its stiffness, you may be able to get by with a five-gallon cooking pot, filled with three gallons of water. A two-pound dress would be better dyed in a pot that holds at least six gallons of water, plus space at the top, so you'd need an eight- to ten-gallon pot. Keep in mind that you should never plan to reuse a dyeing pot for cooking afterwards, because clothing dyes are not considered safe for use on food preparation equipment. A dyeing pot should be made of either stainless steel or of enamel-coated steel; stainless steel is better, but enamel is more economical. You can often buy a 33-quart enamel canner pot for a little under $50, while a stainless steel kettle of the same size will usually cost well over $100.
When you do the dyeing, you will have to stir the dress frequently in the large pot of water, mixed with dye and, for polyester only, an unpleasant smelling intensifier chemical which probably requires you to wear an acid gas respirator for safety. If you don't stir frequently enough, then the color is likely to end up uneven, with some sections darker than others.
Cooking the beads in the dyebath may turn out to be a mistake. They are probably made of plastic. Some plastics will soften at temperatures well below the boiling temperature required to dye polyester, while other plastics are safe up to temperatures over boiling. Unfortunately, it's impossible to determine what the beads on your dress are made of. Even when there is a fiber content label inside the dress at the time of purchase, it will invariably add "exclusive of decoration". The type of plastic used in the beads is never explained. Glass beads will be fine as long as they do not break, though their color will be unchanged by dyeing.
After completing the whole dyeing process, you must then wash the dress to remove any unattached loose dye, so that excess dye doesn't rub off onto you and anything the dress happens to touch. Sometimes a dry-clean-only dress that survives one washing becomes frayed in the next.
Now, let's assume you've done everything right. You've pre-washed your dress, it's turned out not to have any dye-repelling fabric finishes, and you've stirred frequently while boiling the dress in the dye plus chemical additive for an hour. Even after all that, you might find that your results are not good. Sometimes when you dye a commercial garment, one panel of the dress will take the dye much darker than another one does, due to having been cut from a different bolt of fabric. There is no way to predict whether this will happen, except to prevent it by dyeing only PFD ("Prepared For Dyeing") garments that have been sold for the purpose of dyeing. Unfortunately, there is no such thing as a PFD formal dress.
To sum up: you might destroy the dress when washing it; you'll have to spend a lot of money on a dyeing pot; the beading might break, fall off, or melt; and the fabric might not even end up all the same color. I really can't recommend buying a formal dress to dye. There are just too many things that can go wrong.
Instead, I recommend that you try doing more shopping for the dress that is already the right color, or do a web search for "mother of the bride" dresses and look for one that is already black, or hire a local seamstress to make a dress for you in the color that you need.
(Please help support this web site. Thank you.)
Friday, November 18, 2011
Which Dupont dye color is a true black?
Name: Suzanne

Country or region: USA
Message: Which Dupont dye color is a true black?
Unfortunately, the company that manufactures Dupont Silk Dyes is undergoing reorganization. They have discontinued some colors, while they have failed to ship others which they will eventually start to ship again. 700 Noir is among that that Dharma Trading Company does not have in stock now, as of this writing. Surely as basic a color as 700 Noir will not have been discontinued, so Dharma should be able to have it in stock again in the future.
I wonder how their 2019 Noir Concentre compares to the 700 Noir. Dharma does have it in stock. It would be worth testing. The only alternative I know would be to try ordering from one of the firms in Europe that stock H. Dupont dyes, or the California importer Atelier De Paris, hoping that they might still have some in stock from before the reorganization began.
Note that the primaries recommended for mixing your own colors are 393 Canari or 330 Helianthe for yellow; 474 Camelia and 390 Cerisette for red; 108 Bleu Roy or 314 Limoges for blue; and 700 Noir for black.
Thursday, November 17, 2011
Can I dye cotton slipcovers using a cool water dye, so they don't shrink?
Name: Giovanna
Country or region: New York, NY / Rome, Italy
Message: Greetings from NY! You seem like such an authority on the subject of cold water color dyeing, I should like to introduce myself, as a school of architecture drop out with a keen sense of aesthetics and colors, who has gone back to doing what she loves most, interior design work, posing the following problem to you: here I am with two sets of slip covers for my very expensive sofas from Italy, a 100% cotton weave of chenille and threading, currently in a mauvish tone, which no longer works. Replacing them anew would be an even more expensive proposition, was hoping they could be dipped in a black/brown dye, couple local names I consulted with ruled it as impossible, because of the shrinkage factor associated with the dye temperature requirements. but what about cold water color dyeing, might that not be a possibility? Would be grateful for your second opinion and guidance. I would have a large enough swatch for testing.
You certainly should be able to do this with Procion MX fiber reactive dyes, since they react well at temperatures as low as 21°C (or 70°F), though slightly warmer temperatures are preferable.
Normally, we use hot water to wash out the excess unattached dye afterwards, but you can substitute repeated washings at the warmest temperature the fabric can tolerate without shrinking. It's probably best to keep the temperature below 41°C (105°F).
If you have access to a top-loading washing machine, that will be the most convenient way to dye these slipcovers a smooth, even color. (See "How can I dye clothing or fabric in the washing machine?".) If they will not all fit into one load in the washer, then you will have to be very careful to weigh the amount of fabric in each load, and calculate the exact same concentrations of dye, soda ash, and salt per pound of fabric, so that they will all end up the exact same hue.
You can also dye in a five-gallon or larger bucket, but you must stir the fabric in the bath for a long time, ideally an hour. This can be quite tiring, especially if you have to repeat it several times. It is very important to keep the fabric in the dyebath moving, so that you can get a single solid color, instead of a tie-dyed effect.
Alternatively, you can try low water immersion, which is a tremendously easier method that does not require stirring. It will result in variegation of light and dark colors, and usually also of different hues. The results can be very beautiful. Yu can scale this method up to any quantity of fabric. (See "How to Do Low Water Immersion Dyeing".)
Since you are in New York, you can buy your Procion Dyes and soda ash from Aljo Mfg, which has a store located in New York and also sell their dyes by telephone. You will also need a large quantity of ordinary salt. Get some of the water softener sodium hexametaphosphate while you are buying your dyes. There are several other very good sources for ordering these dyes online in the US. I do not know of a source for Procion dyes in Italy, so it is convenient that you are asking this question from New York, instead.
A warning: dyeing will not work well if the fabric has been treated with a stain-resistant finish such as Scotch Guard. You should also know that the stitching that holds together different parts of the slipcovers is almost certainly made from undyeable polyester. Other than that, I think you will have no problems. The fact that your slipcovers are 100% cotton makes this a perfectly reasonable project.
(Please help support this web site. Thank you.)
Wednesday, November 16, 2011
I would like to know the best dyeing procedure in order to salvage my coat.
Name: hkanzki
Country or region: Canada
Message: I have this white winter coat made of a 100% tubular polyster, the exterior is of microporous polyurethane polymerized and the lining is 100% nylon, that I would like to dye into a darker color due to several stains. Please advise me, I would like to know the best dying procedure in order to salvage my coat. I know I am taking a chance, but I consider my coat a loss anyway. Looking forward receiving your advice. Thank you.
I am afraid that your coat is protected with a DWR (durable water repellant) finish, made of either silicone or fluorocarbons. It is usual for any coat which has an polyurethane waterproofing membrane to also be treated with a DWR finish so that the fabric on the outside of the membrane does not become heavy and uncomfortable with absorbed rain water. Any form of waterproofing, including the DWR coating, inevitably makes the coat undyeable, because anything that repels water will also repel dye. The water resistance may be temporarily made ineffective by dirt, but a thorough washing, followed by fifteen minutes in a clothes dryer at medium heat, should restore it to at least partial functionality. (Be sure to remove it from the dryer immediately to avoid permanent creasing.) DWR finishes can never be removed completely enough to allow effective dyeing, but they may need the heat of a clothes dryer to restore their function as much as possible.
Instead of dyeing, you should try more methods to try to remove the stains. If you have not washed the coat with water and detergent, try that first. If cool water doesn't work, try soaking in very hot water. If that's not enough, try soaking it in hot water with a strong oxygen-based cleaner, such as OxyBoost.
After this treatment, including the machine drying step that restores DWR finishes, check to see if the coat still has any water-repellence ability. Sprinkle a few drops of water on the coat (this is to be done only after it is fully dry). If the water beads up, your DWR finish is working well. If there is absolutely no sign that the water is beading up, then perhaps I am mistaken about your coat's having a DWR finish.
None of the above cleaning steps will be wasted, even if you do dye the coat. Even if the coat has no DWR finish, it must still be cleaned as thoroughly as possible before you should even consider dyeing it. Stains always show right through any dye, unless it's a very large amount of dye applied very dark, or unless a variegated-color form of dyeing, such as tie-dyeing or LWI, is used to distract the eye from the former stains. Treatment with several packets at once of Rit Color Remover (a much better product that Rit Dye) in the hottest possible water will take out many dyes and stains, so it's a good step to take before dyeing, if the coat doesn't have any waterproofing at all.
If the coat does have a DWR finish, then the only thing you can do is try to remove stains and renew the finish. I recommend Nikwax TX-Direct products, in either the wash-in or the spray-on forms. You can get these and similar products in other brands from an REI store, or another store that caters specifically to backpackers.
If there is no DWR finish, you can consider dyeing. The combination of polyester and polyurethane is a questionable one for dyeing, unfortunately. Polyester must be boiled in a special polyester dye, because the dye will not work if applied in merely hot water, and no other type of dye will work; it really has to be near boiling. The microporous polyurethane membrane, on the other hand, may be damaged badly by boiling temperatures. Even if damage is not a concern, in order to apply the polyester dye to your coat, you would need a very large cooking pot, one large enough for the coat to move in freely as you stir it in the water. A smaller pot will result in a splotchy or even tie-dyed effect. A sufficiently large cooking pot will be extremely expensive, especially if you follow the rule that says never to reuse a dyeing pot for food. It would probably cost less to buy a new coat.
(Please help support this web site. Thank you.)
Tuesday, November 15, 2011
How to dye 50% Cotton/50% Polyester
Name: **Marisela**
—ADVERTISEMENTS—

Dye polyester and poly/cotton blends
Country or region: USA
Message: Hi, I tried searching "How to dye 50% Cotton 50% Polyester" and found no results. I am emailing you because I have never dyed any clothes before but I have a black sweater that I purchased over 4 years ago and I accidently left in the trunk of a car that was sitting under the sun for 3 years. I just found it and it has faded to a brownish color on the outer part. It is a Carhartt and is made of the following materials: (Outer Shell: 50% Cotton 50% Polyester) & (Inner Lining: 100% Polyester) it weighs 3lbs 4oz. I do not care about the inner lining but it is still black. I do however care about the outer shell. Can you please explain how dyes work, how many bottles I need and what I need to do to make this sweater black again. I truly love this sweater and I would really appreciate your help. I understand there is a risk as I am altering the original fabric but I prefer the risk than this ugly uneven brownish color. Thank you SO MUCH for your time.
Since there is no dye in the world that will work on both cotton and polyester, you will have to use two different dyes. One dye will recolor the cotton fibers in the sweater black, while the other will recolor the polyester fibers black. (The polyester dye will wash out of cotton, while the cotton dye washes out of polyester.)
I do not recommend that you buy Rit brand all-purpose dye from your local store. All-purpose dye does not work very well on cotton, and it does not work at all on polyester. This means that you will probably have to order your dye online. It's worth it, to get better dyes.
Before you go any farther than this, first make sure that the sweater does not have any sort of water-resistant finish. Anything that is water-resistant will also resist dye. When you sprinkle a few drops of water on your sweater, does the water soak in? It's very important to dye only things that can absorb water.
It can be difficult to dye something that is not even in color to start with. Since dye is transparent, the faded portions end up not quite as dark as the other portions, after redyeing. If you use a large enough amount of black dye, though, it will probably cover up the variation. Alternatively you could use Rit Color Remover or Jacquard Color Remover first, to remove as much dye as possible and even out the color somewhat before dyeing. These products remove color without damaging the synthetic fiber the way bleach does.
There are two different approaches to dyeing both the cotton and the polyester. One is to dye the two fibers in separate steps. The other is to buy dyes that can be mixed together and applied at the same time. The dyes that have to be applied separately are better in quality, but the dyes that can be mixed together are less trouble, and they are better than the all-purpose dye mixtures you can buy at the grocery store.
The easy way is to order Jacquard Products' "iDye" and "iDye Poly" in black, and also order an after-treatment called Retayne. "iDye" is a direct dye that will work on cotton, while "iDye Poly" is a completely different kind of dye that will work on polyester. iDye is not the longest-lasting type of dye, but it will last well enough if, after you have completely finished doing your dyeing, you apply Retayne, following the package directions. You can order iDye, iDye Poly and Retayne all at the same time. One supplier that you can order iDye and iDye Poly from is Blick Art Materials ; Dharma Trading Company sells both of these dyes, as well as many others, and also sells Retayne. You will need to actually cook the sweater on your stovetop in a big pot along with both of these dyes; it's not enough to just use hot water. The pot must be large enough for the sweater to move freely in the water as you stir it. ; Dharma Trading Company sells both of these dyes, as well as many others, and also sells Retayne. You will need to actually cook the sweater on your stovetop in a big pot along with both of these dyes; it's not enough to just use hot water. The pot must be large enough for the sweater to move freely in the water as you stir it.
Alternatively, you can order Procion MX dye for dyeing the cotton part of your fiber blend, and another brand of disperse dye for dyeing the polyester. I like to order disperse dyes from either PRO Chemical & Dye or Aljo Mfg, both of which, like Dharma, also sell Procion MX dyes. Procion MX dyes are among the very best and longest-lasting dyes for cotton, and they don't require heat or a cooking pot to use. They do require soda ash or washing soda to act as a dye fixative. For a beginner, doing your dyeing in a single step is less trouble, and you'll still have to boil the sweater in dye in order to dye the polyester in it.
Retayne is an easy way to make direct dyes like iDye more permanent on cotton. You can apply it by stirring a spoonful of it with the sweater in a bucket of very hot water. It also works for all-purpose dye, but, if you're going to the trouble of ordering Retayne online, you may as well order a better dye than Rit, especially since you'll almost certainly need to order online to get polyester dye.
(Please help support this web site. Thank you.)
Monday, November 14, 2011
I have a silk and guipure lace white wedding dress I would like to dye black to make into an evening gown
Name: Stephanie
Country or region: Hertfordshire, England
Message: I have a silk and guipure lace white wedding dress I would like to dye black to make into an evening gown. Can you suggest what dye I should use or is there a place where I could get this professionally done?
Is your dress washable? Have you washed it yet? It seems likely that your guipure lace should not be washed, for fear of shrinkage and puckering of the lace. If a dress is not washable, then it simply cannot be dyed, either at home or commercially. For more on that, see my blog entry from August 18, 2011.
If your dress has been washed without any problems, then it might be dyeable, but the fact is that you will be taking some risk with this. A small but significant fraction of items will be ruined when you try to dye them; they may prove to be less sturdy than expected and fall apart in the wash, or it may turn out that one panel of fabric is different from the others and ends up taking a different amount of the dye, resulting in a noticeable difference in color. I recommend dyeing used or other non-PFD* clothing only if you can accept that there is some risk of a total loss.
Guipure lace is not made of silk, so it may not be easy to get a perfect color match between the dress and the silk. Different types of fibers typically produce slightly different hues with the same dye. Some guipure lace is made of 100% rayon (viscose rayon, not rayon acetate), and is therefore easily dyeable, though the silk may perhaps dye to a browner or bluer or more greenish shade of black than the rayon. Worse, some guipure lace contains a little polyester and nylon; without unpicking the lace from the silk before dyeing, nothing that is part polyester can be made to dye as dark a shade as the silk, since polyester requires more heat for dyeing than silk can be expected to survive.
If the dress is washable, and if the lace is 100% rayon instead of 10% polyester, then there are several possible choices of dyes. You might get good enough results by using several packets of an all-purpose dye (one packet is unlikely to produce a dark black) or by using Dylon Machine Dye, which contains a fiber reactive dye. (Dylong Machine Dye is available in the UK and the rest of Europe, but not in North America.) It's important to pre-wash the dress very thoroughly before attempting to dye it.
I can't tell you what professional dyers there may be in England, as none have ever written to me asking to be listed on my "Find a Custom Dyer" page, and nobody who's written to me has ever mentioned success with one. All of the custom dyers on my list are in the US or Canada.
(Please help support this web site. Thank you.) *PFD means clothing that is commercially Prepared For Dyeing; it is made of easily dyeable material, often sewn together with dyeable cotton thread instead of the standard polyester thread, and free of finishes that can inhibit dyeing.
Sunday, November 13, 2011
Do you have any suggestions on the best way to dye ivory lace to gray?
Name: Sandra
Country or region: USA
Message: Hi. I recently purchased 64 yards of Ivory lace to make table runners for my wedding. After purchasing now I wish I had gone with gray!! So do you have any suggestions on the best way to dye ivory lace to gray? I won't / or don't need to dye the 64 yards all together, the plan was to after my aunt cuts the lace into the size each one needs to be to make the table runners we were going to dye each one indvidually but I'm scared of it ruining the lace and then i'm out the $$ I spent on the lace and also no table runners. We are going to cut a swatch and play with it but came across this so thought I would ask :o)
What is the fiber content of the lace? It's absolutely vital to know the fiber content, before you can even consider dyeing. Different fibers require completely different dyes!
If you can't find out the fiber content from the retailer or manufacturer, try a burn test (carefully!). Two excellent resources for interpreting the results of a fiber burn test are Griffin Dyeworks' Burn Test page and Ditzy Prints' Fiber Burn Chart.
After you learn what fiber your lace is composed of, we can discuss how you can dye it, or possibly even whether you should dye it at all, depending on the fiber content.
(Please help support this web site. Thank you.)
Saturday, November 12, 2011
How much soda ash should I use to scour fabric before dyeing?
Name: Leslie
—ADVERTISEMENTS—


Country or region: East coast USA
Message: Hi, Paula--
I've tried searching for the answer to this, believe me...
I want to scour a small amount of PFD cotton (3 yards), and wondered if I can do it in our big utility tub with very hot water, Synthrapol and soda ash. If so, what would be the amount of soda ash to add to the water? Is there a rule of thumb for soda ash per gallon of water for smaller quantities of fabric, like, say, per yard? Or is the amount of soda ash the same as in the cold-water immersion process per gallon? I really don't want to put such a small amount of fabric in my washer, which doesn't do such small loads....
The purpose of the scouring step is to clean your fabric before dyeing. It helps in removing invisible sizing or other finishes that can interfere with dyeing, though there are some finishes that can't be removed so easily. Don't confuse the soda ash used in scouring with the soda ash used in fixing the dye. The soda ash used in the scouring stage will be rinsed out, so it won't do anything to help fix your dye.
The very best way to pre-scour fabric, before dyeing, is to boil the fabric on the stovetop. This is more effective than pre-scouring at the cooler temperatures that are all you can manage in a washing machine. You should not use an aluminum pot for this step, because aluminum reacts badly with soda ash. A stainless steel or enameled pot is ideal.
If you don't want to use a cooking pot for your scouring, just use your utility tub, a sink, or a large bucket, with the hottest water you can get. In very cold weather, you may want to warm the tub first with hot water, drain it, and then scour your fabric with more hot water. You will need to stir or agitate the fabric repeatedly in the water, along with the soda ash and Synthrapol. If you're going to get your hands in the water, be sure to wear rubber gloves, such as the ones used for dishwashing, that are a long enough to protect your wrists, because you don't want to get soda ash on your hands unnecessarily, and, ideally, the water should be scalding hot anyway.
PRO Chemical & Dye says to use half a teaspoon (2 gm) of soda ash and half a teaspoon (2.5 ml) of Synthrapol for scouring each pound of fabric; they don't specify how much water to use, but it should be a large enough amount that the fabric can move freely in it. It's really not critical exactly how much soda ash you use. It's best to weigh your fabric, while it's still dry, to know how much you have, but they give a rough estimate that one pound of fabric equals 3 to 4 yards of cotton muslin. Of course, heavier fabrics weigh more per yard.
After scouring your fabric, drain off the water, which may be visibly dirty depending on the cotton, and rinse with clean water. You can use the spin cycle of some washing machines to remove excess water at this step, without having to fill the washing machine or run a full cycle.
(Please help support this web site. Thank you.)
Thursday, November 10, 2011
What is the purpose of using sodium sulfate, when dyeing fabrics with Procion MX dyes?
Name: Jennifer
Country or region: Florida
Message: What is the purpose of using sodium sulfate, when dyeing fabrics with Procion MX dyes? I have seen instructions both with and without using it. If I decide to use it, do different products cause different results?
 This is really two questions: 1, what is the purpose of using salt when dyeing with Procion dyes, and 2, what is the difference between Glauber's salt (sodium sulfate) and regular salt (sodium chloride), when dyeing with Procion dyes. This is really two questions: 1, what is the purpose of using salt when dyeing with Procion dyes, and 2, what is the difference between Glauber's salt (sodium sulfate) and regular salt (sodium chloride), when dyeing with Procion dyes.
The first question is covered in my FAQ page, "Do I need to use salt, in dyeing?" . Whether you need to use any salt at all, including sodium chloride or sodium sulfate, depends on what kind of dyeing you are doing. You do not need to add any salt when you are tie-dyeing or dye-painting, because the dyes being used are so concentrated. You do need to add salt to your dyebath for high water ratio immersion dyeing, as when dyeing in a washing machine or a five-gallon bucket, to prevent dye from being wasted in the large volume of water; using a large amount of salt helps keep the negative charges of the dyes and the fiber from causing them to repel each other. Using salt in your high-volume dyebaths helps to increase the color yield you get from a given amount of dye.
You don't need to add salt to dye concentrates used for tie-dyeing or dye painting because the total volume of water is so low, relative to the amount of dye and fiber, that the dye manages to find the fiber anyway. In addition, adding salt to dye concentrates can be a problem, because salt reduces the solubility of dye. If the dye in your mixture becomes insoluble, dropping to the bottom of the container in the form of powder, because the addition of too much salt has reduced its ability to dissolve in water, then your colors will end up very pale. I've had this happen to me, so I recommend against adding salt to your dye concentrates.
For low water immersion (LWI) dyeing, the amount of water used is small enough that your results will be good and intense even without salt. However, adding salt to your low-water dyebath will tend to increase the sharpness of the crystalline-like patterns that are formed in LWI. Don't add the salt directly to your dye concentrates; instead, add a strong solution of salt to your fabric. I like to add salt to my LWI baths along with the soda ash, after the dye has been soaking into the fabric for at least a few minutes.
The second question is, why use sodium sulfate, instead of sodium chloride? Sodium sulfate is more expensive, after all. Sodium sulfate and sodium chloride will do pretty much the same thing in a fiber reactive dyebath. Why not use the cheaper and easier-to-buy sodium chloride?
Although both are salts, and the two have a lot in common, sodium sulfate has some different properties than sodium chloride when used with acid dyes. Sodium sulfate slows the attachment of acid dye to fiber via the sulfate groups in the dye molecules, and thus aids in getting a more level, even color, but that's not an issue when working with fiber reactive dyes. Reactive dyes don't bond to fibers via their sulfate groups, the way acid dyes do.
The one reason I've seen for recommending sodium sulfate instead of sodium chloride, with reactive dyes, is that it is supposed to intensify the color of copper phthalocyanine based dyes, such as Procion Turquoise MX-G. For example, Dharma Trading Company says in their catalog,
"Improves the yield of Turquoise Fiber Reactive Dye. (Gives more intense color). Use in place of,and in the same proportions as, Plain Salt, when dyeing solid shades of #25 Turquoise or colors mixed with #25 (marked with T on the color chart)." (There's more information on this particular dye on the page "A Beautiful Blue: Procion Turquoise MX-G".) I don't know why this particular dye structure is supposed to be more affected by sodium sulfate than other dye colors are. Having something improve the yield of one dye color more than other dye colors could wreak havoc with color mixing, if not enough testing is done first.
If you're dyeing something a pure turquoise, using Procion turquoise blue MX-G, for example, then, according to this recommendation, you might get a more intense turquoise color, for a given amount of dye, if you substitute sodium sulfate for sodium chloride, or perhaps you can get away with using a smaller amount of turquoise dye, for the same color intensity. As with sodium chloride, you wouldn't want to do this with tie-dyeing or dye painting, because of the solubility problem, but you could use it in solid-color dyeing, or in low water immersion dyeing.
If you are dyeing anything with a mixture of dye colors, however, including not only Procion MX turquoise MX-G but also at least one other color of dye, then using sodium sulfate could alter the balance of color, and therefore your hue. If you are using a pre-mixed color, or a color recipe you've perfected without the use of sodium sulfate, then sodium sulfate will make the final result more blue in color, if it's true that sodium sulfate acts this way. Substituting sodium sulfate for sodium chloride might change a green mixture to a bluish one. Note that of the hundred or so colors sold by the best dye retailers, such as Dharma or ProChem, only a dozen or so are pure single-hue dyes; the others are mixtures of two or more dye colors.
Substituting sodium sulfate for sodium chloride makes a great deal of sense to try if you're dyeing several tons of garments the exact same color, because the trial and error of getting the exact right hue, by varying the amount of each dye you use, would be well worth the effort in order to reduce the cost required in buying large quantities of dye.
If you're doing very small-scale hand dyeing, though, it makes much less sense to even try this. If you buy premixed colors of Procion MX dye, you expect your result to end up similar in hue to the color chip in the catalog of your supplier. If you have already worked out recipes to mix your own colors, using sodium chloride in your dyebaths, then your colors might change when you substitute sulfate for chloride. It might not be worth all the trial and error of figuring out exactly how much less turquoise dye powder you can get away with using, by substituting sodium sulfate for sodium chloride.
(I should point out that I have not experimented myself to see how big a difference in dye yield there is for turquoise as opposed to other colors of dye. Turquoise is noted for suffering more from a cold studio temperature than other colors do, since it reacts more slowly than they do; is it possible that this is the only reason why sodium sulfate is supposed to make a bigger difference in color yield for turquoise-based dyes than for other colors? Be sure to include notes on temperature when you do your own tests.)
I recommend using sodium sulfate as a substitute for sodium chloride with Procion MX dyes only when you are experimenting. It's always wise to do tests before any large or important project, anyway. Changing from one salt to another is fine if you do a good series of test swatches first to make sure what color you will get, and it's also fine if you're not testing at all but will be happy with the unexpected. If you have some sodium sulfate on hand anyway, and you're doing some low water immersion dyeing, go ahead and try sodium sulfate to see what happens. Don't do it if you're not willing to risk getting a different hue than you expect.
Interestingly, sodium sulfate is commonly used to dilute dye powders to a standard strength per gram. The same type of dye from one retailer may contain sodium sulfate, while that from another retailer may contain a synthetic tannin as a diluent powder, instead. (See my page, "What is in dye powder?".) Colorado Wholesale Dyes makes a point of selling dyes that don't have extra salts added to them as diluents; they think this makes it easier to wash out excess dye after dyeing, but it could also make a difference in the reproducibility of colors you mix yourself. The presence of salt certainly does make a difference in how much dye you can dissolve in a certain amount of water.
(Please help support this web site. Thank you.)
Monday, November 07, 2011
Can I make Procion MX an acid dye for silk so I can use the serti technique with gutta, dry and steam?
Name: Bonny
Country or region: Canada
Message: Can I make Procion MX an acid dye for silk so I can use the serti technique with gutta, dry and steam? If I use vinegar instead of water for the dilutant. Thank you for this terrific web site. It has helped me through many trials.
Yes, you can use Procion MX dyes as acid dyes on silk. Procion MX dyes work pretty well as acid dyes, when used with an acid instead of with soda ash. (See "Fiber reactive dyes on protein fibers".)
Instead of using vinegar as a presoak, you may prefer to follow PRO Chemical & Dye's recipe for doing this with citric acid, which does not evaporate the way vinegar does. If you use vinegar in your dye mix, then allow the dye to dry thoroughly before steaming as is usual, then the vinegar will evaporate, too, along with the water, but citric acid does not evaporate.
A good recipe for doing this is ProChem's Silk Painting using PRO MX Reactive Dyes [PDF]. It calls for Procion MX dye, urea, citric acid crystals, Synthrapol, and your choice of gutta or silk resist. It doesn't call for steaming, but you will probably find that your colors are better if you do steam anyway, when using Procion MX dyes as acid dyes.
Citric acid can often be found locally, in the form of 'sour salt' for cooking, or in a store that sells supplies for home wine-making. You can also purchase it from a good dye supplier, such as G&S Dye in Toronto. It's easy to ship, because it is in the form of a dry powder. See my page about citric acid, if you'd like to know more about it, though you really don't need to do anything more than follow the recipe mentioned above.
It would make sense to also include Ludigol in your dye mixtures. Ludigol improves the color yield of Procion type dyes when they are steamed, by preventing the dye from being reduced at high temperatures, which tends to bleach out the dyes. ProChem sells Ludigol under the name "PRO Chem Flakes", while G&S Dye sells it as "Ludigol" and Dharma Trading Company sells it as "Ludigal F". Use one level tablespoon, or 8 grams, per quart or liter of chemical water mixture; that's the same as 3/4 teaspoon, or two grams, per 250-ml cup.
The recipe also calls for Synthrapol, the detergent, which is available only from dye suppliers. G&S TNA Soap is a perfectly good substitute. See "What is Synthrapol?".
The urea called for in the recipe should not be necessary at all, if you choose to steam your silk after the dye paint has dried on it. (See "What is urea for? Is it necessary?".) The primary purpose of urea in dyeing with Procion MX dye is to retain moisture, especially when dyeing with soda ash or another high-pH chemical, to allow plenty of time for the dye reaction to occur before the dye dries out. Wrapping the dyed material in plastic, as specified in this recipe, makes the humectant properties of urea unnecessary. However, if you will be steaming your painted silk, after it dries, there will be no need to slow its drying time. In some cases, urea increases the solubility of dyes to a small extent, so that you can dissolve more in the same amount of water, but this is unlikely to be an issue for you. If you will be steaming your dye-painted silk, feel free to omit the urea from the recipe, or try it with the urea one time, and without it another. Retaining extra moisture in the dye paint might sometimes increase the amount that the color spreads. This won't matter if your gutta lines are very well applied.
Be sure to use plenty of newsprint paper to wrap your painted silk in for steaming, to prevent the dye from one part of the silk from transferring onto another. The newsprint that touches your silk should be either unprinted white newsprint paper, or it should be aged at least six months after printing, in order to avoid transfer of the newspaper ink to your silk.
Another way to use Procion MX dyes for painting silk with the serti technique is to use a regular tie-dyeing recipe to prepare your dyes, mixing your dye paints with soda ash immediately before use. The usual tie-dye dye concentrates made with Procion MX dyes work well when used as thin paints for direct application. If you would like to thicken your dyes, see Michael Fowler's recipe on my page, "Sodium alginate, Superclear, and other dye thickeners". This method calls for soda ash, even though it can also be used on silk without any changes. Although it is made of protein, silk is more resistant to damage from high pH than wool and other protein fibers are. Since Procion MX dye bonds well to silk at room temperature, when used with soda ash, there is absolutely no need to steam when following this protocol. Urea is a very helpful ingredient with this technique.
(Please help support this web site. Thank you.)
Sunday, November 06, 2011
I am trying to locate a vendor for 45" shoe laces that I can dye.
Name: Laborde
Country or region: US
Message: I am trying to locate a vendor for 45" shoe laces that I can dye. Dharma is all out. Any suggestions? Thank you for your help.
I can't vouch for how well they dye, but there appear to be some other sources for 100% cotton showlaces. See these two, for example:
Dharma Trading Company is supposed to get theirs back in stock in a couple of weeks.
Avoid waxed laces, because the wax will prevent the dye from reaching the cotton.
(Please help support this web site. Thank you.)
Wednesday, November 02, 2011
Is it possible to permanently UV dye a climbing rope?
Name: Jim
Country or region: UK
Message: Please can you help. I want to UV permanently dye "climbing rope". I would like to know how permanent does "permanent" mean. I know this may be a shot in the dark idea, but if you could help I would be most grateful.
Can you tell me what the rope is made of? Nylon can be dyed pretty well, but polypropylene is hopeless and cannot even be painted.
What do you mean by "UV permanently dye"? Are you talking about a fluorescent dye, which absorbs ultraviolet light and then re-emits the energy as visible light?
Thanks very much for your e-mail. I am pretty sure the rope is nylon. What I want to do is immerse the rope in a UV dye solution for a period of time, allow it to dry out and to be able to "see" the rope using a black UV light source.
If this feasible then what sort of dye and colour would be the most appropriate and how long would the dye last for if often used ?
Hoping you can help.
"Black light" is ultraviolet light, which is invisible to our eyes. The dyes and pigments that absorb black light and then emit light that we can see are called fluorescent.
It's not impossible to find a good fluorescent dye for nylon (in fact, it's much easier than finding one for cotton), but, unfortunately, the absorption of the energy in ultraviolet light makes all of these dyes very susceptible to light damage. There is no such thing as a truly permanent fluorescent dye. Your ropes will have to be re-dyed after too much exposure to bright light. I very much doubt their brightness would survive even a month left exposed to daily bright sun all day long, maybe not even a week. Be sure to store them in a dark place when they are not being displayed or used.
Note that some nylons have chemical coatings that prevent dyes from getting into the fiber. Always test-dye a small sample before spending too much time or money on a project.
You can't dye nylon by immersing it in room-temperature dye. The best dyes for Nylon dyes are acid dyes, which require heating. It's not enough to start with hot water in your dyebath; you must do your dyeing in a cooking pot, so that you can maintain the high temperature for some time. Don't use an aluminum cooking pot for this purpose, since aluminum reacts badly with the mild acids (such as vinegar) used along with acid dyes, and don't plan to reuse your dyeing pot for food preparation, because some fluorescent dyes are somewhat toxic and might be mildly carcinogenic, and no real textile dyes have been cleared for safety when used in food. Be careful to keep the temperature of your dyebath no higher than 96°C (which is 205°F), and use something in the bottom of your pot as a spacer to prevent your nylon from contacting any hot spots where the metal is thinner on the bottom of your dyeing pot. A hot spot may damage and weaken the nylon.
Where to buy fluorescent acid dyes for nylon in the UK? I'm finding it difficult to find more than one or two colors in your country. The dye supplier George Weil sells Jacquard Acid Dyes, but there's only one fluorescent acid dye in that line of dyes, the hot pink Rhodamine B (also known as Colour Index Acid Red 52, or Jacquard's 620 Hot Fuchsia). Like all fluorescent textile dyes, its lightfastness is poor, so it will fade relatively quickly in bright sunlight.
Flavine Yellow is another fluorescent acid dye, sold in the US by PRO Chemical & Dye, which should be available elsewhere. Its generic name is Colour Index Acid Yellow 7, and ProChem's name is Washfast Acid 107A Flavine Yellow G. A web search for ("acid yellow 7" AND UK) turns up some possible sources, such as WA Products (UK) Ltd. You would do best to look up the recipe for ProChem's "Immersion Dyeing on Nylon using WashFast Acid Dyes" [PDF], as the instructions given by WA Products are for an entirely different use. Note that, again, as with all true fluorescent dyes, acid yellow 7 fades quickly in bright sunlight.
The best source I know for multiple colors of fluorescent dyes for nylon is Aljo Mfg, in New York, in the US. Although you can examine their line of products online, they require orders to be made by telephone or by mail. It's worth it, since they carry more colors of fluorescent true dye. Look under their listings for "silk/wool" dyes, rather than for "acetate/nylon" dyes; nylon actually dyes with better washfastness with the "silk/wool" acid dyes than with the "acetate/nylon" disperse dyes, and the "silk/wool" line includes several true fluorescents, Fluorescent Rhodamine B, Fluorescent Blue G, Fluorescent Violet, and Fluorescent Flavine Yellow. I do not know of any other source for Fluorescent Blue G and Fluorescent Violet acid dyes. Other fluorescent dyes I can see advertised for sale elsewhere online include acridine orange, which I showed in my PhD research to cause DNA damage by means of the photodynamic effect, which means that it's somewhat likely to cause cancer, and rhodamine G, which is toxic and probably carcinogenic; I do not recommend hand-dyeing with these two dyes.
As an alternative, consider fluorescent fabric paint. It's not as good as a fluorescent dye, but it is easier to acquire in a variety of colors. Fabric paint will always wear off much more quickly than a true dye, because it adheres only to the outside of each fiber, instead of soaking through. Some fabric paints will alter the feel of the rope more than others, but, unlike true dyes, all fabric paints will affect it some, which could be a real issue if the rope will ever be used for climbing. It might be worth a try, however, depending on the uses to which the rope will be put.
With luck, you may find your fluorescent paint to be more resistant to light-fading than the fluorescent dyes, though I can't guess how much longer they may last. The longest-lasting color in fluorescent nylon will involve solution dyeing, which requires the addition of fluorescent pigments to the liquid nylon, before it is extruded into fibers; unfortunately, that's not an option for you, but do check carefully to see whether there is solution-dyed fluorescent nylon rope on the market already.
If you use a Jacquard Products brand paint, such as the fluorescent yellow, fluorescent orange, fluorescent pink, fluorescent red, fluorescent blue, fluorescent green, and fluorescent violet colors of Jacquard Textile Colors (if George Weil doesn't have them, they should be able to special-order them for you), you can avoid the necessity of heat-setting by buying some Jacquard Airfix; adding Airfix to a fabric paint immediately before use allows the acrylic in the paint to cure without ironing or other heat-setting steps, which will make the whole project much easier. Another UK source for a variety of fluorescent paints appears to be UV Gear.
Always test any product you are considering on a small, discardable piece of similar rope before deciding whether it will work well for you. If you are planning to use the rope for actual climbing, I strongly recommend that you have the rope properly tested for strength after you have dyed it; it's not impossible that the heat of dyeing might have bad effects on the strength of the rope, which is something you certainly do not want to learn while climbing! Name: Rebecca
Country or region: Netherlands
Message: Hi there, I've got a gorgeous vintage felt hat which is a vibrant and bright orange. I'd like to dye this hat olive green or grey or black but definitely NOT brown. I have no additional material to do color tests on. I've had some "unexpected" results in the past by dyeing colorful items with black dye and I'm wondering of you know of any color charts which can help me better control/anticipate the outcome. I'd be willing to go for deep purple or a dark burnt orange or another color if I could just predict a bit better how this color and material would react to the dyeing process. Many thanks in advance, any tips/advice/links will be most greatly appreciated!
A bright orange seriously limits you in what colors you can change it to. You'll never be able to overdye a very bright orange to change it to any sort of purple or green. Purple can't have the yellow in it that orange has, while green can't have the red in it. I don't think a neutral gray of any sort will be possible, either.
The choices you have, starting from a bright orange, are burnt orange, red, dark red, brown, or black.
If you use brown dye or brown fabric paint, you should be able to turn a bright orange to a burnt orange. A pure magenta dye will make it red, while a true red will make an orangish red. A medium blue will create a brown color. If you use a lot of brown dye, the hat will be brown, and black dye should result in a brownish black. A blue black should result in a true black.
A nice way to visualize color mixing is with the Dye Mixer Applet, created by Olli Niemitalo, at http://iki.fi/o/dye/dyemixer/ . Choose an orange dye using one slider bar, then use another slider bar to add on another dye color. You can quickly see what colors work and what colors do not work. It won't tell you the correct dyes to use for your hat, but it will show you what sort of color you want to use.
Another very important question is, what is the fiber content of your hat? It won't do you any good to choose the right color, if the dye you choose does not work. Wool felt or fur felt should be dyed with acid dyes. Acid dyes will not work on synthetics (except for nylon). Dyeing with acid dyes requires heat and plenty of water; the hat will lose its shape and will need to be re-blocked to restore its shape. You might also consider using a thin fabric paint, such as Dye-Na-Flow. Most fabric paints, and all thin ones, are transparent, so you should expect a fabric paint to follow the exact same rules of color mixing as for dye.
(Please help support this web site. Thank you.)
|
|















 This is really two questions: 1, what is the purpose of using salt when dyeing with Procion dyes, and 2, what is the difference between Glauber's salt (sodium sulfate) and regular salt (sodium chloride), when dyeing with Procion dyes.
This is really two questions: 1, what is the purpose of using salt when dyeing with Procion dyes, and 2, what is the difference between Glauber's salt (sodium sulfate) and regular salt (sodium chloride), when dyeing with Procion dyes.







